 :max_bytes(150000):strip_icc()/alkalineearth-56a12cd75f9b58b7d0bcca7e.png)
Radium, barium, and strontium have few industrial applications, while magnesium and calcium have many uses in manufacturing and industry.Radium was discovered by scientists Marie and Pierre Curie.English chemist Sir Humphry Davy was the first to isolate many of the alkaline earth metals including calcium, strontium, magnesium, and barium. 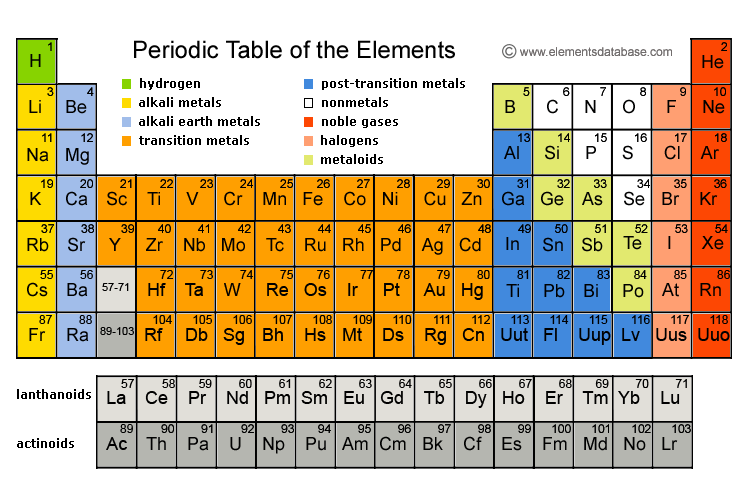
Calcium plays an important role in helping us to build strong bones and magnesium is used to help regulate the body's temperature. Calcium and magnesium are important for animal and plant life.It is very radioactive and is dangerous to handle. Radium is formed from the decay of uranium.They are called alkaline because they form solutions with a pH greater than 7, making them bases or "alkaline." The name "alkaline earths" comes from an old name for the oxides of the elements.The elements in group 1 are known as the alkali metals those in group 2 are the alkaline earth metals those in 15. They burn with various colored flames as follows: beryllium (white), magnesium (bright white), calcium (red), strontium (crimson), barium (green), and radium (red). Groups are numbered 118 from left to right.Interesting Facts about Alkaline Earth Metals It is the fifth most abundant element in Earths crust. Its physical and chemical properties are most similar to its heavier homologues strontium and barium. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. The most abundant of the alkaline earth metals on Earth is calcium which is the fifth most abundant element in the Earth's crust. Calcium is a chemical element with the symbol Ca and atomic number 20. They tend to form ionic bonds, except for beryllium which forms covalent bonds.All of them except beryllium react strongly with water.They react with halogens to form compounds called halides.They all occur in nature, but are only found in compounds and minerals, not in their elemental forms.They have two outer valence electrons which they readily lose.They are fairly reactive under standard conditions.They are silvery, shiny, and relatively soft metals.What are the similar properties of alkaline earth metals?Īlkaline earth metals share many similar properties including: Click the links or see below for more details on each. The elements of the alkaline earth metals include beryllium, magnesium, calcium, strontium, barium, and radium. They are sometimes referred to as the group 2 elements. They are all in the second column of the periodic table. It is a tabular arrangement of the elements by their chemical properties that usually uses abbreviated chemical symbols in place of full element names, but the linear list format presented here is. are oxidized) when they undergo chemical reactions They normally do not accept electrons. It is the sixth element in group 2 of the periodic table, also known as the alkaline earth. The alkaline earth metals are a group of elements in the periodic table. Electropositive Character: Metals tend to have low ionization energies, and typically lose electrons (i.e. Radium is a chemical element with the symbol Ra and atomic number 88. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
